PC4-4
Effect of co-substitution on superconductivity in Y1-xCaxBa2-ySryCu4O8 prepared by the molten KOH method
Dec.2 10:15-10:30 (Tokyo Time)
Department of Applied Physics, Tohoku University, Sendai, Japan1
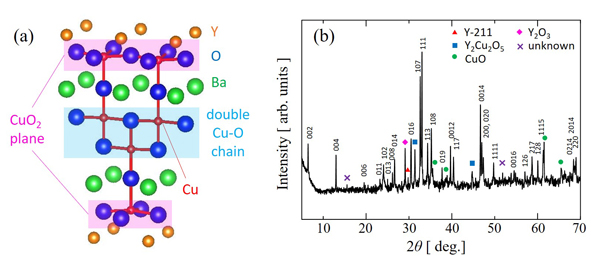
The cuprate superconductor YBa2Cu4O8 (Y-124) has the double Cu-O chains without oxygen nonstoichiometry as shown in Fig. 1(a), while YBa2Cu3O7-δ (Y-123) has the single Cu-O chains with oxygen nonstoichiometry. Due to this high stability of oxygen content, Y-124 is a promising material for the practical application of superconductivity. However, Tc ~ 80 K of Y-124 is too low for the practical application at 77 K. The relatively low Tc of Y-124 is due to the low hole-concentration in the CuO2 planes. It is reported that its Tc increases up to ~90 K, which is almost the same as 92 K of Y-123, by increasing the hole concentration through the partial substitution of Ca2+ for Y3+ [1]. More interestingly, Tc is greatly enhanced up to 108 K by application of a high pressure of 12 GPa [2]. On the other hand, Tc increases only up to about 90 K by applying the chemical pressure through the partial substitution of Sr2+ for Ba2+ [3]. In this study, we have investigated the effects of co-substitution of Ca2+ for Y3+ and Sr2+ for Ba2+ on Tc in Y1-xCaxBa2-ySryCu4O8 to explore the possibility of Tc ~ 100 K at ambient pressure.
Samples of Y1-xCaxBa2-ySryCu4O8 were prepared by the molten KOH method. As shown in Fig. 1(b), Y-124 phase was obtained for x(Ca) ≤ 0.2 and y(Sr) ≤ 0.2, although small amounts of impurity phases are included.We will report the co-substitution effect on Tc.
[1] T. Miyatake et al., Nature 341, 41(1989). [2] E. N. van Eenige et al., Physica C 168, 482 (1990). [3] Y. Miyachi et al., Jpn. J. Appl. Phys. 58, 070906 (2019).
Fig. 1 (a) Crystal structure of Y-124. (b) Powder x-ray diffraction pattern of Y0.8Ca0.2Ba1.8Sr0.2Cu4O8.
Keywords: Y-124, elemental substitution, superconducting transition temperature, hole concentration